|
The NADH decrease is directly proportional to the ALT activity.(Package insert: ALT reagent. Roche Diagnostics 04/2019)Īlanine aminotransferase (ALT) activity is determined by a kinetic method using a coupled enzyme reaction where the rate of NADH consumption is measured at 340 nm. It is determined by measuring the increase in absorbance.(Package insert: Alkaline Phosphatase Gen.2 reagent. The p-nitrophenol released is directly proportional to the catalytic alkaline phosphatase activity. In the presence of magnesium and zinc ions, p-nitrophenyl phosphate is cleaved by phosphatases into phosphate and p-nitrophenol. Roche Diagnostics 02/2022)Īspartate aminotransferase (AST) is measured by a coupled enzyme kinetic method where the rate of decrease of NADH, determined at 340 nm, is directly proportional to the AST activity.(Package insert: AST reagent. The color intensity of the blue-green albumin-BCG complex is directly proportional to the albumin concentration and is determined photometrically.(Package insert: Albumin Gen.2 reagent. The dye, bromcresol green (BCG), is added to serum in an acid buffer. The color intensity is directly proportional to the protein concentration, which can be determined photometrically.(Package insert: Total Protein Gen.2 reagent, Roche Diagnostics 11/2019) Sodium potassium tartrate prevents the precipitation of copper hydroxide and potassium iodide prevents autoreduction of copper. Roche Diagnostics 11/2021)ĭivalent copper reacts in alkaline solution with protein peptide bonds to form the characteristic purple-colored biuret complex. The rate of NADPH formation is directly proportional to glucose concentration and is measured photometrically.(Package insert: Glucose HK Gen.3 Reagent. In the presence of nicotinamide adenine dinucleotide phosphate (NADP+), glucose-6-phosphate dehydrogenase oxides G6P to gluconate-6-phosphate and NADPH. Glucose in the serum, in the presence of hexokinase, is converted to glucose-6-phosphate (G6P). The change in absorbance is directly proportional to the calcium concentration and is measured photometrically.(Package insert: Calcium Gen.2 reagent. This complex reacts in the second step with EDTA. Roche Diagnostics 02/2019)Ĭalcium ions react with NM-BAPTA (5-nitro-5'-methyl-1,2-bisethane-N,N,N',N'-tetraacetic acid) under alkaline conditions to form a complex. The color intensity of the quinone imine chromogen formed is directly proportional to the creatinine concentration in the reaction mixture.(Package insert: Creatinine. Catalyzed by peroxidase the liberated hydrogen peroxide reacts with 4-aminophenazone and HTIB (hydroxyiodobenzene) to form a quinone imine chromogen. This enzymatic method is based on the conversion of creatinine with the aid of creatininase, creatinase, and sarcosine oxidase to glycine, formaldehyde, and hydrogen peroxide. The decrease in absorbance, due to the consumption of NADH, is measured kinetically and is proportional to the amount of urea in the sample.(Package insert: Urea/BUN reagent. The ammonia formed then reacts with alpha-ketoglutarate and NADH in the presence of urease/glutamate dehydrogenase to yield glutamate and NAD+. This is a kinetic UV assay where urease cleaves urea to form ammonia and carbon dioxide. The following equation is used to calculate the anion gap (A gap): 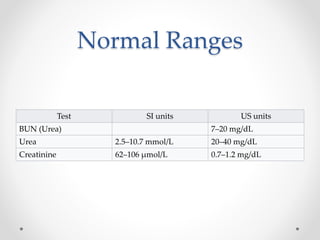
The rate of change is directly proportional to the concentration of bicarbonate.(Package insert: Bicarbonate reagent Roche Diagnostics 04/2019) The consumption of NADH causes a decrease in absorbance and is monitored in the UV range of 320 nm to 400 nm. The oxaloacetate produced is coupled with NADH in the presence of malate dehydrogenase (MDH) to produce malate and NAD. Bicarbonate (HCO3-) reacts with phosphoenolpyruvate (PEP) in the presence of phosphoenolpyruvate carboxylase (PEPC) to produce oxaloacetate and phosphate. An electronic calculation circuit converts EMF of the sample to the ion concentration of the sample.(Package insert: ISE Indirect Na, K, CL for Gen2. 
The EMF of the reference electrode is constant. The ion selective electron (ISE) module indirectly measures the electromotive force (EMF) difference between an ISE and a reference electrode. The EMF of the ion-selective electrode is dependent on the ion concentration of the sample. Ion-selective electrode (ISE) (indirect potentiometry).  
Reference values have not been established for patients who are younger than 12 months of age.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
